Product and Vendor News
Product Business Settings
Certara GlobalSubmit eCTD software is popular in Pharmaceuticals industry and is widely used by Enterprise.
Certara GlobalSubmit eCTD software Product Overview
Globalsubmit provides eCTD submission software to mitigate rejection risks in a complex regulatory environment. Differences in regulatory requirements complicate the submission process.
How satisfied the customers are with Certara GlobalSubmit eCTD software use-cases
Reviews
"...GlobalSubmits approach to document management is a familiar one intuitive interface, easy implementation that appeals to the novice user, built-in templates and workflows, and integration with eCTD publishing systems...." Peer review
Certara GlobalSubmit eCTD software Customer Insights, Testimonials and Case Studies
Certara GlobalSubmit eCTD software Features
- Low
- Medium
- High
| FEATURE | RATINGS AND REVIEWS |
|---|---|
| Custom Reports | Read Reviews (48) |
| Analytics | Read Reviews (1) |
| CAPABILITIES | RATINGS AND REVIEWS |
|---|---|
| Custom Reports | Read Reviews (48) |
| Analytics | Read Reviews (1) |
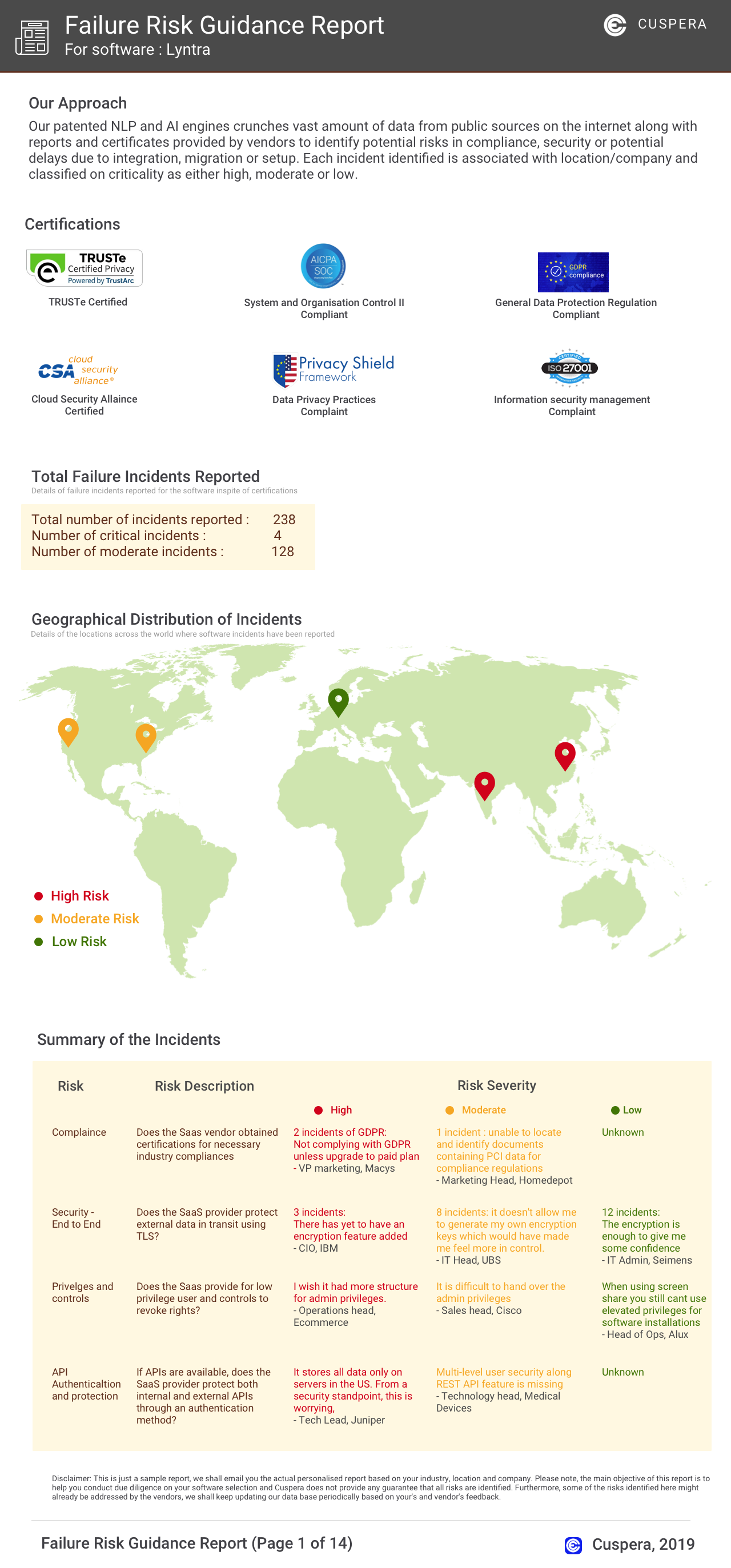
Software Failure Risk Guidance
?for Certara GlobalSubmit eCTD software
Overall Risk Meter
Top Failure Risks for Certara GlobalSubmit eCTD software
Certara, USA. Profile
Company Name
Certara, USA.
Company Website
https://www.certara.com/HQ Location
123 South Broad Street, Suite 1850, Philadelphia, PA 19109, US
Employees
11-50
Social
Financials
PRIVATE